Air Quality Lab
Collaborators- Sam Freeman
Abstract- To test air quality, we used a mixture of several substances on a sheet of paper to measure the amount of ozone in the air and smeared petroleum jelly on another to see the amount of particulate matter. The controls for this lab were placed inside in isolation, while the experimental were placed outside. After 24 hours, we compared the two. Through these methods, we observed that the air quality was relatively good with not too much ozone and little particulate matter.
Problem- What is the air quality of Wake Forest in terms of ozone and particulate matter?
Hypothesis- (ozone) If we measure the ozone of inside and outside, the outdoors will have higher amounts because there is more exposure to sources of heat, sunlight, exhaust fumes and other sources.
(PM) If we measure the amount of particulate matter of inside and outside, the outdoors will have higher amounts because there are not as many sources of PM that are indoors.
Parts of Experiment- Control group- Isolated ozone measurement and indoor particulate matter.
Experimental group- Outdoor measurements of ozone and PM.
Independent variable- Where we place the measurements outside.
Materials- Test Tube Rack
Filter Paper
Microscope slides
Petroleum Jelly
Q-tips
Microscopes
Potassium Iodide
Corn starch
Beakers
Spatulas
Large Graduated Cylinder
Methods- (Ozone) 1. Get 100ml of water and put it in a 250 ml beaker, and add 5g of cornstarch. Then heat and stir the mixture until it gels. Remove the solution from heat, add 1 g of potassium iodide, stir well, and allow to cool
2. Soak two pieces of paper in the resulting substance.
3. Hang one piece of paper outside, preferably out of direct sunlight, and the other inside inside a plastic bag out if sunlight.
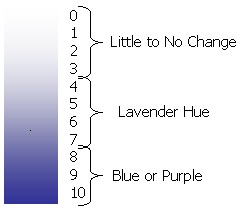
4. Wait 24 hours and compare the two pieces of paper according to this scale:
Abstract- To test air quality, we used a mixture of several substances on a sheet of paper to measure the amount of ozone in the air and smeared petroleum jelly on another to see the amount of particulate matter. The controls for this lab were placed inside in isolation, while the experimental were placed outside. After 24 hours, we compared the two. Through these methods, we observed that the air quality was relatively good with not too much ozone and little particulate matter.
Problem- What is the air quality of Wake Forest in terms of ozone and particulate matter?
Hypothesis- (ozone) If we measure the ozone of inside and outside, the outdoors will have higher amounts because there is more exposure to sources of heat, sunlight, exhaust fumes and other sources.
(PM) If we measure the amount of particulate matter of inside and outside, the outdoors will have higher amounts because there are not as many sources of PM that are indoors.
Parts of Experiment- Control group- Isolated ozone measurement and indoor particulate matter.
Experimental group- Outdoor measurements of ozone and PM.
Independent variable- Where we place the measurements outside.
Materials- Test Tube Rack
Filter Paper
Microscope slides
Petroleum Jelly
Q-tips
Microscopes
Potassium Iodide
Corn starch
Beakers
Spatulas
Large Graduated Cylinder
Methods- (Ozone) 1. Get 100ml of water and put it in a 250 ml beaker, and add 5g of cornstarch. Then heat and stir the mixture until it gels. Remove the solution from heat, add 1 g of potassium iodide, stir well, and allow to cool
2. Soak two pieces of paper in the resulting substance.
3. Hang one piece of paper outside, preferably out of direct sunlight, and the other inside inside a plastic bag out if sunlight.
4. Wait 24 hours and compare the two pieces of paper according to this scale:
Methods- (PM) 1. Smear thin layers of petroleum jelly onto two note cards.
2. Hang a note card outside and the other one inside.
3. Wait 24 hours and compare the two using your eyes and microscope and look for any particulate matter inside the petroleum jelly.
2. Hang a note card outside and the other one inside.
3. Wait 24 hours and compare the two using your eyes and microscope and look for any particulate matter inside the petroleum jelly.
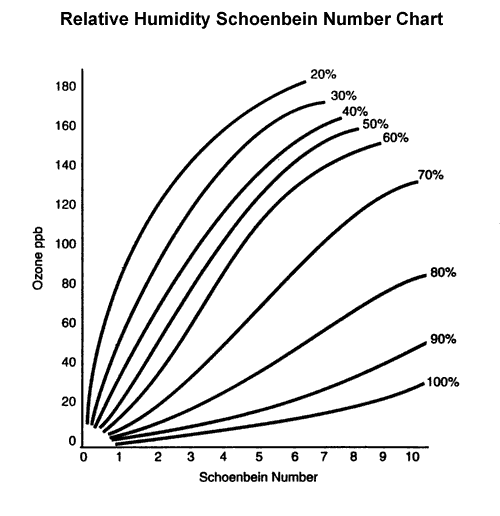
Data- (Ozone) Our outdoor ozone paper came back with a darker lavender hue which meant the ozone was a 6-7 on the scale with a humidity of about 50%, it was about 150 ppb. The indoor one, on the other hand, came back completely white which meant there was no change at all.
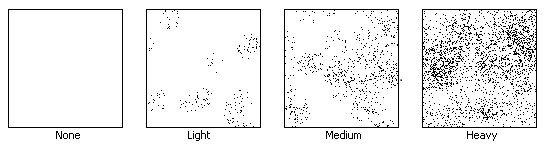
(PM) Our outdoor PM paper came back with very few particulate matter on it. Very few specs here and there, a few insects and small spots which we speculate to be particles of dust or dirt. The indoor one came back with almost nothing at all on it. Using the microscope, we spotted small particles of dust, but nothing more.
Data Analysis- (Ozone) When comparing our results for the outdoor ozone level, we noticed that ours was relatively high. Many of our peers got ozone ppb that were much lower than what we collected. This could have been for several reasons. One could have been that our paper was in direct sunlight for an amount of time. Sunlight and heat cause ozone levels to rise, so that could have happened to ours. Another possibility could have been that since we placed our paper near a parking lot, the emissions from all the vehicles could have caused the ozone levels to rise.
(Particulate Matter) Our data was relatively the same compared to others. Our area has low levels of PM in the air. One factor that could have affected us, though, was that we placed our note cards high up off of the ground, so it is possible that less particulate matter got onto it because it was so high up.
Lab Questions-
1. The highest rating the EPA will allow for a community for ground level ozone is 80 ppb. Any community with over 80 ppb for an average over three years is in noncompliance with the Clean Air Act. Compare your number to this standard. Did Wake Forest meet the standard on this day? Using what you know about the formation of ozone, decide if you believe the measurement you made was an accurate measurement of the air quality and why.
Our results said that the ozone ppb of Wake Forest was about 150. If this were true, Wake Forest would not have net the standards. However, we might have placed the paper in direct sunlight and it affected our results, causing the ozone measurement to skyrocket.
2. 13% of counties in the US are not meeting the Ozone requirements of the Clean Air Act. Use the chart below and information from class to explain the reason for the location of the counties in Non-attainment.
The counties that are not meeting the ozone requirements are highly populated areas with large cities or communities that would make it hard to meet the regulations of the Clean Air Act.
3. Particulate matter has recently been added to the requirements of the Clean Air Act. Theorize reasons that it was not part of the act until the 1990’s.
Maybe it wasn't considered much of a problem, and since many types of PM were so common, maybe they thought it would be too troublesome to enforce. Also, according to the Air Quality Management Division, PM wasn't considered too dangerous until the last decade, when they discovered even smaller PM particles that can cause lots of respiratory damage(1).
4. Compare and contrast the different PM samples – did they meet your expectations? Why or why not?
I was actually expecting a bit more PM from the outdoor sample, seeing as it was outdoors. I didn't expect our air to be as relatively clean as it is.
5. Explain why air quality monitoring is important for the health of the economy.
Air quality monitoring is good for the economy because if someone is working outside in poor air quality, he could get injured or even die. The company could be sued for making this person work in poor conditions. So, knowing the air quality is very important to the health of the economy.
Conclusions- Our results for the ozone supported our hypothesis with it having higher ozone levels than the control. The outdoors had higher ozone with the amount of sunlight and heat, however, the paper must have been in direct sunlight, causing it to record much higher ozone levels than in reality. Our results for particulate matter somewhat did support our thesis statement. The outdoor PM sample had a bit more particles than the indoor one, but not by much.
Citations-
1. "Particulate Matter: Little things can cause big problems." Hamilton County
Environmental Services: Air Quality Management Division. N.p., n.d. Web. 19
Sept. 2013. <http://www.hcdoes.org/airquality/monitoring/pm.htm>.
(PM) Our outdoor PM paper came back with very few particulate matter on it. Very few specs here and there, a few insects and small spots which we speculate to be particles of dust or dirt. The indoor one came back with almost nothing at all on it. Using the microscope, we spotted small particles of dust, but nothing more.
Data Analysis- (Ozone) When comparing our results for the outdoor ozone level, we noticed that ours was relatively high. Many of our peers got ozone ppb that were much lower than what we collected. This could have been for several reasons. One could have been that our paper was in direct sunlight for an amount of time. Sunlight and heat cause ozone levels to rise, so that could have happened to ours. Another possibility could have been that since we placed our paper near a parking lot, the emissions from all the vehicles could have caused the ozone levels to rise.
(Particulate Matter) Our data was relatively the same compared to others. Our area has low levels of PM in the air. One factor that could have affected us, though, was that we placed our note cards high up off of the ground, so it is possible that less particulate matter got onto it because it was so high up.
Lab Questions-
1. The highest rating the EPA will allow for a community for ground level ozone is 80 ppb. Any community with over 80 ppb for an average over three years is in noncompliance with the Clean Air Act. Compare your number to this standard. Did Wake Forest meet the standard on this day? Using what you know about the formation of ozone, decide if you believe the measurement you made was an accurate measurement of the air quality and why.
Our results said that the ozone ppb of Wake Forest was about 150. If this were true, Wake Forest would not have net the standards. However, we might have placed the paper in direct sunlight and it affected our results, causing the ozone measurement to skyrocket.
2. 13% of counties in the US are not meeting the Ozone requirements of the Clean Air Act. Use the chart below and information from class to explain the reason for the location of the counties in Non-attainment.
The counties that are not meeting the ozone requirements are highly populated areas with large cities or communities that would make it hard to meet the regulations of the Clean Air Act.
3. Particulate matter has recently been added to the requirements of the Clean Air Act. Theorize reasons that it was not part of the act until the 1990’s.
Maybe it wasn't considered much of a problem, and since many types of PM were so common, maybe they thought it would be too troublesome to enforce. Also, according to the Air Quality Management Division, PM wasn't considered too dangerous until the last decade, when they discovered even smaller PM particles that can cause lots of respiratory damage(1).
4. Compare and contrast the different PM samples – did they meet your expectations? Why or why not?
I was actually expecting a bit more PM from the outdoor sample, seeing as it was outdoors. I didn't expect our air to be as relatively clean as it is.
5. Explain why air quality monitoring is important for the health of the economy.
Air quality monitoring is good for the economy because if someone is working outside in poor air quality, he could get injured or even die. The company could be sued for making this person work in poor conditions. So, knowing the air quality is very important to the health of the economy.
Conclusions- Our results for the ozone supported our hypothesis with it having higher ozone levels than the control. The outdoors had higher ozone with the amount of sunlight and heat, however, the paper must have been in direct sunlight, causing it to record much higher ozone levels than in reality. Our results for particulate matter somewhat did support our thesis statement. The outdoor PM sample had a bit more particles than the indoor one, but not by much.
Citations-
1. "Particulate Matter: Little things can cause big problems." Hamilton County
Environmental Services: Air Quality Management Division. N.p., n.d. Web. 19
Sept. 2013. <http://www.hcdoes.org/airquality/monitoring/pm.htm>.